Articles
November 16, 2023
Adding (Ultra-) Rare Disease Products to Your Company’s Portfolio: It’s a Whole New “Ball Game” (Part 1)
Many advanced pharmaceutical and biotech companies are moving into (ultra-) rare disease markets. High levels of unmet need are translating into steadily growing R&D expenditures and increasing revenues in rare diseases (RDs), so interest is not expected to wane any time soon.
Figure 1 – Key figures on the European Medicines Agency’s (EMA) recommendations for the authorisation of new medicines in 2022; Source: EMA report, Human Medicines Highlights 2022, page 1, published 2023.
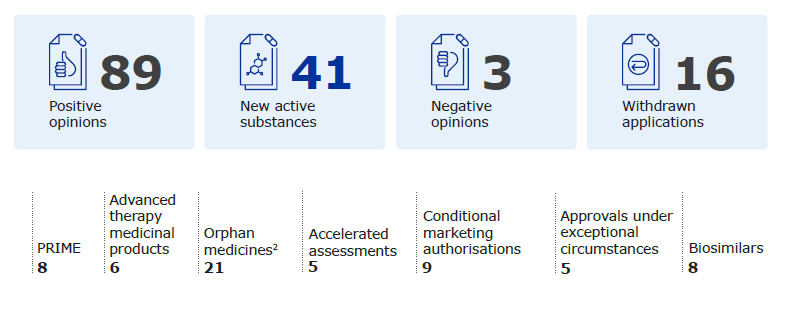
Additionally, one-product RD companies are increasingly partnering with—or are acquisition targets for—midsize and larger pharmaceutical companies, who are interested in expanding their portfolios into markets with smaller patient populations. The increased focus on this space is all good news, especially for patients who suffer from RDs. However, there is a potential catch when it comes to the commercial distribution of these new products.
In this short article (part 1 in a 2-part series), we describe the special challenges associated with distributing RD products. Next week, in part 2, we will outline the key steps and deliverables for developing and implementing supply chains for RD therapies. In addition, we will summarize some of the pitfalls that biopharma companies must avoid when working to establish reliable supply chains in rare diseases.
What’s the Catch?
Globally, biopharmaceutical supply chains and commercial distribution processes have generally been designed to meet the requirements of medium- to high-volume products for larger patient populations, with (relatively speaking) medium to low prices and requiring standard conditions for their storage, handling, and distribution. This typically involves maintaining temperatures of 2-8 °C or 15-25 °C, the use of conditioned trucks and/or (more than) pallet-sized active or passive cooling solutions, and handling systems targeted towards volume rather than value.
When it comes to RD products, however, the characteristics and requirements can be far from what most would consider normal. They often involve:
- Low volumes for low numbers of patients
- High values
- Highly specific—and often challenging—requirements for storage, handling, and distribution, often with (temperature excursion) specifications that are much “tighter” than traditional norms
- Special requirements when it comes to Patient Support and/or Home Care Services, customer training and certifications, etc.
In addition, dosage forms may be non-standard, and prescription, treatment and reimbursement processes may require specific or additional steps to be taken. Besides all that, the distribution channels for RD products are generally more focused on Direct-to-Hospital, Direct-to-Pharmacy, and Direct-to-Patient models (the latter sometimes in combination with Home Care solutions and/or Patient Support Services necessary for providing the treatment). A consequence of using these channels is that sufficient access to “local languages” in the selected customer service model is often of key importance, as professional command of the language considered to be “international” (English) at the customer side is not always a given.
The combination of distribution characteristics and channel choices typically leads to limited (or no) stocks being held in the channels or being present at the point of use. The amount of working capital locked up, the high storage cost, the risk of shelf life expiry, and high potential write-off costs are the main drivers of keeping such limited stocks. This makes it much more challenging for companies to meet target delivery performance levels and to achieve reliable fulfillment without the implementation of RD-focused solutions.
While the above list of challenges is not exhaustive, it does show that adding RD products to a company’s existing portfolio can be a very complicated endeavor. For any given product, it requires an in-depth assessment and analysis to determine the “best fitting” supply chain distribution model.
Coming Next
To date, AIM has helped more than 75 companies design and build supply chain and operations infrastructures for RD products. By using the insights gained from those experiences, we’ve evolved a multi-step process to help decision-makers identify and address the critical aspects of supply chain development for therapies with such unique and/or specialized requirements. In part 2, we provide a high-level description of the key steps, activities, and deliverables in this process.